Introduction to Chemical Bonding
Chemical bonding is a fundamental concept in chemistry that describes the forces holding atoms together in a molecule. It is essential to understand chemical bonds as they dictate how atoms interact, combine, and form various compounds. At the heart of chemical bonding are two primary types: ionic and covalent bonds, each featuring distinct characteristics and implications for the properties of the resulting substances.
Chemical bonds arise from the interactions between the electrons of atoms. When atoms approach each other, they may share or transfer electrons, resulting in stable arrangements. Ionic bonds occur when electrons are transferred from one atom to another, leading to the formation of charged ions that attract each other. Conversely, covalent bonds are formed when atoms share electrons, allowing them to attain greater stability. Understanding these interactions is crucial since the properties of compounds—such as boiling and melting points, solubility, and reactivity—are directly influenced by the type of bonds present.
AD
The significance of chemical bonding extends beyond mere atomic interactions; it is integral in determining material properties and behaviors in various contexts. For instance, the hardness of diamonds is a result of the strong tetrahedral covalent bonds between carbon atoms, while the malleability of metals can be attributed to the presence of metallic bonding. As compounds exhibit various physical and chemical characteristics dictated by their bond types, insights into chemical bonding provide a lens through which scientists can predict behavior and reactivity.
Types of Chemical Bonds
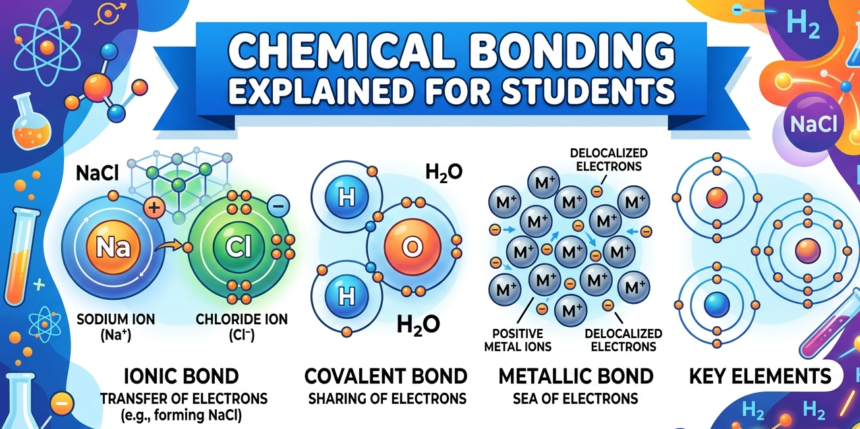
Chemical bonds are fundamental interactions between atoms that enable the formation of compounds. The three primary types of chemical bonds are ionic, covalent, and metallic bonds. Each type exhibits unique characteristics and forms based on the nature of the atoms involved.
Ionic bonds occur when electrons are transferred from one atom to another, typically between metals and nonmetals. In this process, the metal atom loses one or more electrons to become a positively charged ion (cation), while the nonmetal gains those electrons to become a negatively charged ion (anion). The electrostatic attraction between these oppositely charged ions results in the formation of ionic compounds. A classic example of an ionic bond is sodium chloride (NaCl), commonly known as table salt, where sodium donates an electron to chlorine.
Covalent bonds, on the other hand, involve the sharing of electron pairs between atoms. This type of bond predominantly forms between two nonmetals. In covalent bonding, each atom contributes at least one electron to the shared pair, creating a strong connection. The number of shared electron pairs determines whether the bond is single, double, or triple. Water (H2O) and carbon dioxide (CO2) are crucial examples of molecular compounds formed through covalent bonding.
Lastly, metallic bonds consist of a shared pool of electrons that are free to move around metals. In this bond type, positively charged metal ions are surrounded by a sea of delocalized electrons, which facilitates conductivity and malleability. This characteristic explains why metals such as copper and aluminum can conduct electricity efficiently and be molded into various shapes without breaking.
Understanding these types of chemical bonds is crucial in chemistry, as they form the basis for how elements interact and combine to create diverse materials and compounds.
Ionic Bonds Explained
Ionic bonds are a fundamental type of chemical bond that occurs when electrons are transferred from one atom to another, resulting in the formation of ions. This process generally happens between metals and nonmetals due to significant differences in their electronegativities. Electronegativity is the measure of an atom’s ability to attract and hold onto electrons. In ionic bonding, metals, which have low electronegativity, tend to lose electrons, while nonmetals, possessing higher electronegativity, accept these electrons.
The formation of ionic bonds involves two key concepts: ionization energy and electron affinity. Ionization energy is the energy required to remove an electron from a neutral atom, converting it into a positively charged ion or cation. For example, sodium (Na), a metal with low ionization energy, readily loses one electron to achieve a stable electronic configuration, forming the Na+ ion. Conversely, electron affinity is the energy change associated with the addition of an electron to a neutral atom, transforming it into a negatively charged ion or anion. Chlorine (Cl), a nonmetal with high electron affinity, readily gains an electron to form Cl–.
As these oppositely charged ions, the cation and anion, come together, they experience an electrostatic attraction that results in the creation of an ionic bond. Ionic compounds, such as sodium chloride (NaCl), exemplify this bonding type. In NaCl, the sodium ion (Na+) and the chloride ion (Cl–) bond to form a stable compound, reflecting the underlying principles of ionic bonding.
In summary, ionic bonds are characterized by the complete transfer of electrons between atoms, highlighting essential concepts such as electronegativity, ionization energy, and examples like sodium chloride that exhibit this bonding behavior.
Covalent Bonds Explained
Covalent bonds are a fundamental type of chemical bond formed through the sharing of electrons between two or more atoms. This type of bonding often occurs between nonmetals, which have similar electronegativities. The motivation behind the sharing of electrons is to achieve a full outer shell of electrons, leading to enhanced stability for the involved atoms, as described by the octet rule.
There are several types of covalent bonds, classified primarily based on the number of shared electron pairs. A single covalent bond involves the sharing of one pair of electrons, for instance, in the molecule of hydrogen (H2). In this case, two hydrogen atoms share a single pair of electrons, resulting in a stable diatomic molecule. A double covalent bond occurs when two pairs of electrons are shared, exemplified by the oxygen molecule (O2). This bond strengthens the interaction between the two oxygen atoms, making it more robust than a single bond. A triple covalent bond, characterized by the sharing of three pairs of electrons, is observed in nitrogen (N2). This type of bond is particularly strong and significant in biological and chemical processes due to its stability and energy requirements for breaking.
Another aspect of covalent bonds is bond polarity, which arises when the electronegativity of the atoms involved in the bond is different. In such cases, the shared electrons tend to be drawn closer to the atom with the higher electronegativity, resulting in a partial charge. This leads to polar covalent bonds, as seen in water (H2O), where the oxygen atom holds a partial negative charge, while the hydrogen atoms acquire a partial positive charge. Conversely, in a nonpolar covalent bond, as found in methane (CH4), the sharing of electrons occurs equally between similar atoms, resulting in no charge separation.
Overall, covalent bonds play a crucial role in the formation of numerous compounds essential for life and various chemical reactions. Understanding the nature and characteristics of these bonds provides insight into the behavior of molecules and the principles governing chemical interactions.
Metallic Bonds Explained
Metallic bonding is a type of chemical bond that occurs between metal atoms. It is characterized by the sharing of free electrons among a lattice of metal cations. This phenomenon can be described using the concept of a ‘sea of electrons,’ where the valence electrons of metal atoms are delocalized and free to move throughout the structure, unlike the localized electrons found in covalent or ionic bonds.
This unique arrangement not only contributes to the distinctive properties of metals but also underlies their functionality in various applications. One of the hallmark characteristics of metallic substances is electrical conductivity. The free electrons allow electrical current to flow easily through the metal, making them excellent conductors. This feature is critical for metals used in electrical wiring, electronics, and numerous engineering applications.
Furthermore, the malleability and ductility of metals are also attributed to metallic bonding. When a metal is subjected to stress, the layers of cations can slide over each other without breaking the metallic bond. This allows metals to be easily hammered into sheets (malleability) or drawn into wires (ductility). For instance, gold and copper exhibit exceptional malleability and are commonly utilized in jewelry and electrical components, respectively.
Additionally, metals tend to have high melting and boiling points, resulting from the strong attractive forces between the delocalized electrons and the positively charged metal ions. Examples of metallic substances include iron, which is extensively used in construction, aluminum, known for its lightweight properties, and titanium, cherished for its strength-to-weight ratio. The understanding of metallic bonds is crucial for various scientific and industrial fields, as they form the foundation for many materials utilized in everyday life.
Bond Strength and Energy
Bond strength refers to the measure of the energy required to break a chemical bond, reflecting how strongly atoms are held together within a molecule. Various types of chemical bonds exist, including ionic, covalent, and metallic bonds, each having distinct bond strengths. For instance, covalent bonds typically exhibit higher bond strengths than ionic bonds due to the sharing of electron pairs between atoms, which results in a more stable configuration.
The energy associated with a bond is termed bond energy, often quantified in kilojoules per mole (kJ/mol). This metric signifies the amount of energy needed to break one mole of a specific bond in a molecule. High bond energy indicates a strong bond, while low bond energy suggests weaker interactions that can more easily be disrupted. To measure bond energy, scientists often analyze the enthalpy changes during a chemical reaction. By comparing the total bond energies of reactants and products, they can infer the energy required for bond formation or breaking.
Several factors influence bond strength and energy. These include atomic size, electronegativity, and the nature of the bond (e.g., single, double, or triple). As atomic size increases, bond strength generally decreases because the distance between bonded nuclei becomes larger. Furthermore, electronegativity plays a crucial role; bonds formed between atoms with significant differences in electronegativity tend to be stronger due to increased ionic character.
Bond length, the distance between the nuclei of bonded atoms, is inversely related to bond strength. Typically, shorter bonds are stronger than longer ones because the closer proximity of nuclei enhances nuclear attractions. This relationship highlights the interplay between bond strength, bond length, and bond energy, illustrating fundamental concepts vital for understanding chemical bonding.
Polarity of Bonds
The polarity of chemical bonds is a fundamental concept in understanding molecular behavior and reactivity. Polarity arises from the differences in electronegativity between two atoms that are covalently bonded. Electronegativity refers to the ability of an atom to attract electrons toward itself in a bond. When two atoms have significantly different electronegativities, the bonding electrons tend to be drawn closer to the more electronegative atom, resulting in a polar bond.
For instance, in a water molecule (H2O), oxygen is more electronegative than hydrogen, causing the electrons to spend more time orbiting the oxygen atom. This unequal sharing of electrons leads to a partial negative charge near the oxygen and a partial positive charge near the hydrogen atoms, illustrating a polar covalent bond. The polarity of water is critical as it influences various properties, such as its solvent capabilities and surface tension.
On the other hand, when two atoms have similar electronegativities—like the two carbon atoms in ethane (C2H6)—the electrons are shared equally, resulting in a nonpolar bond. In nonpolar covalent bonds, the symmetrical distribution of electron density ensures that there are no regions of partial charge, giving rise to a molecule with a uniform charge distribution.
The implications of bond polarity extend beyond simple classification into polar or nonpolar. For example, polar molecules often engage in dipole-dipole interactions and hydrogen bonding, while nonpolar molecules may interact through dispersion forces. Understanding the polarity of bonds is essential for predicting molecular behavior in various chemical reactions and environments.
The Role of Chemical Bonding in Molecular Geometry
Chemical bonding plays a critical role in determining molecular geometry, which refers to the three-dimensional arrangement of atoms within a molecule. Understanding how different types of chemical bonds affect the shape of a molecule is essential in both theoretical and practical chemistry. One of the key theories utilized to explain molecular shapes is the Valence Shell Electron Pair Repulsion (VSEPR) theory. This theory posits that the geometric arrangement of electron pairs around a central atom dictates the overall shape of the molecule.
According to VSEPR theory, electron pairs—whether bonding pairs or lone pairs—repel each other due to their negative charges. This repulsion leads to a specific spatial arrangement that minimizes the energy of the molecule. For instance, in a molecule like methane (CH4), the four hydrogen atoms repel each other equally, resulting in a tetrahedral geometry. Similarly, in water (H2O), the two hydrogen atoms and two lone pairs of electrons around the oxygen create a bent shape. Such spatial arrangements have profound implications for the physical and chemical properties of substances.
The influence of molecular geometry derived from chemical bonding impacts various properties, including polarity, reactivity, and interactions with other molecules. For example, the shape of a molecule can determine whether it is polar or non-polar, affecting its solubility and intermolecular interactions. Consequently, the understanding of molecular geometry is vital for predicting the behavior of compounds in chemical reactions. Therefore, the essential nature of chemical bonding extends beyond mere atom connection; it is intricately linked to the structure and behavior of molecules in scientific studies and applications.
Conclusion and Importance of Understanding Chemical Bonding
In summarizing the various aspects of chemical bonding, it is crucial to highlight its fundamental role in the field of chemistry. Chemical bonding forms the basis for the interactions between atoms, which subsequently leads to the formation of molecules. Understanding the types of bonds—ionic, covalent, and metallic—provides students with insights into how substances behave in different environments. This foundational knowledge is not merely academic; it has practical applications that permeate various aspects of everyday life.
For students, grasping the principles of chemical bonding is essential for predicting how different materials will react, which in turn impacts areas such as pharmacology, materials science, and environmental science. By studying these interactions, students can better understand phenomena such as the structure of enzymes and the properties of polymers, which hold significant implications for both industrial applications and research advancements.
Furthermore, a solid comprehension of chemical bonding principles fosters critical thinking and problem-solving skills. As students engage with complex concepts in chemistry, they develop a rigorous approach to analyzing and interpreting scientific data. This analytical foundation is invaluable in various scientific disciplines, thereby expanding their career opportunities in research, healthcare, and engineering fields.
Overall, the importance of understanding chemical bonding cannot be overstated. It not only enriches students’ knowledge but also equips them with the necessary tools to tackle real-world challenges. By delving into the world of chemical interactions, students prepare themselves to contribute effectively to scientific advancements and innovations, ultimately shaping the future of technology and sustainability.